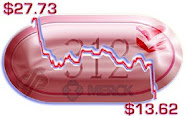
These candidates will -- if ultimately approved -- enter as "me too" competitors to Gilead's front running Solvadi®. It should also be noted that Solvadi will likely have cut its prices wildly, in the developing world (reducing Merck's opportunities there) -- and with Congressional committees pounding on pricing here, it won't be a pretty picture at home. At least not likely to be an easy $100,000 plus per patient juggernaut, now -- not for the "me too" entrants.
This is good news for Merck -- make no mistake. It is also good news that Merck is entering Phase III with at least one study here. It may just not be materially good news, given Merck's overall size. In any event, here is a bit of the press release, this morning:
. . . .MK-5172 an investigational HCV NS3/4A protease inhibitor and MK-8742 an investigational HCV NS5A replication complex inhibitor – are scheduled to be presented at the 49th Annual Meeting of the European Association for the Study of the Liver (EASL) also known as The International Liver Congress™ 2014. The data are from Merck’s overall Phase 2 clinical program. The meeting will take place in London United Kingdom April 9 - 13 2014.
Based on the results of the Phase 2 program Merck is initiating a Phase 3 clinical trial program to be named C-EDGE. The C-EDGE program is designed to evaluate these investigational treatments across genotypes and in different HCV subpopulations including patients with chronic kidney disease HIV/HCV co-infection and cirrhosis. . . .
As I say, this is a big market, but Gilead has, and will have, a likely multi-year "first mover" advantage, in Hep C, by the time these two clear all the regulatory baffles. It will help Merck that it will have HIV/HCV co-infection data in its favor when approved. But that won't necessarily make it a mono-therapy, first line treatment, even in that arena. Do stay tuned.















1 comment:
Note that the data about Solvaldi's effectiveness is nowhere near as positive as how it's been spun. See:
http://hcrenewal.blogspot.com/2014/03/too-good-to-be-true-sovaldi-kerfuffle.html
Post a Comment